
Lavanya
The European Union has finalized one of its most consequential chemical substance bans in recent years. Regulation (EU) 2024/1849, amending the original EU Mercury Regulation (EU) 2017/852, mandates a total phase-out of dental amalgam for general use and bans the manufacture, import, and export of six additional categories of mercury-containing lamps. For manufacturers, importers, and healthcare suppliers operating in EU markets, understanding the EU mercury regulation 2026 compliance requirements is no longer optional — it is an immediate operational imperative.
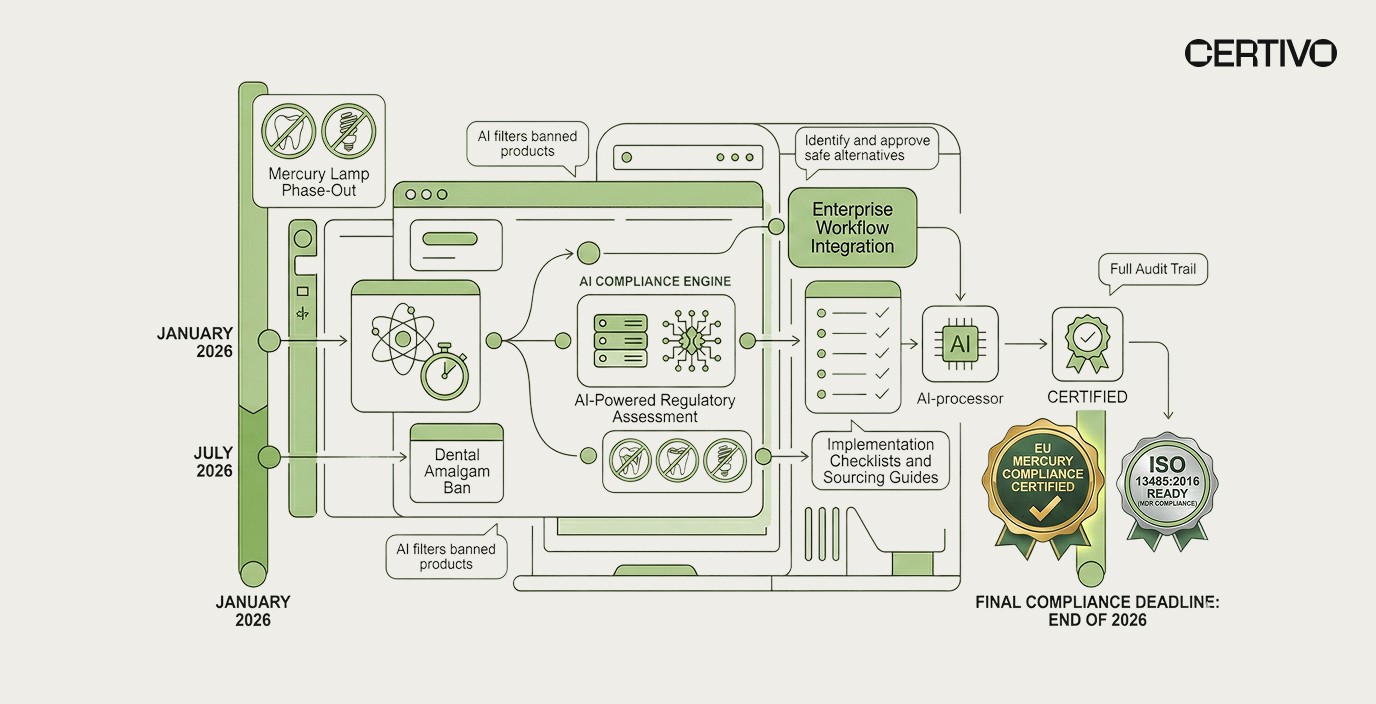
The regulation's phased deadlines have already begun. The general ban on dental amalgam use and exports took effect on January 1, 2025. The next wave — covering mercury lamp manufacturing and export bans — hits December 31, 2025, with final restrictions on dental amalgam manufacturing, import, and remaining lamp categories closing by December 31, 2026. CEOs and executive leadership must treat this as a board-level compliance priority.
This article provides a complete breakdown of every obligation, deadline, affected industry, and compliance action required under the amended regulation.
Table of Contents
What Changed: The Scope of Regulation (EU) 2024/1849
Key Requirements and Obligations
Phased Compliance Deadlines: Full Timeline
Affected Industries and Products
Dental Amalgam Ban: Detailed Obligations
Mercury Lamp Ban: Detailed Obligations
Compliance Risks and Penalties
Operational and Supply Chain Impact
Compliance Readiness Checklist
The Role of AI in Mercury Compliance Monitoring
FAQs
Conclusion
1. What Changed: The Scope of Regulation (EU) 2024/1849
The original EU Mercury Regulation (EU) 2017/852 already restricted certain uses of mercury. However, it permitted continued use of dental amalgam and allowed several categories of mercury-containing lamps to remain on the market. The 2024 amendment eliminates those remaining carve-outs entirely.
The stated policy objective is to eliminate the last intentional uses of mercury across the EU. This aligns with the EU's Zero Pollution Action Plan and its international commitments under the Minamata Convention on Mercury. The regulation accelerates the transition to mercury-free alternatives — composite resins in dentistry and LED technology in lighting. Organizations tracking EU chemical policy shifts should review the broader context of key EU regulations manufacturers must watch.
⚠ Critical scope note: The regulation applies across all EU Member States, including Northern Ireland under the Windsor Framework. Any entity manufacturing, importing, exporting, or using dental amalgam or mercury-containing lamps within this jurisdiction is in scope.
📊 What's new in the 2024 amendment:
Total ban on dental amalgam use for general patients (limited medical-need exceptions only)
Prohibition on exporting dental amalgam to third countries
Prohibition on manufacturing and importing dental amalgam into the EU (from July 1, 2026)
Ban on manufacture and export of six additional categories of mercury-containing lamps
Temporary derogation for Member States with specific healthcare reimbursement needs (expiring June 30, 2026)
For compliance and regulation managers overseeing EU market access, this regulation introduces hard stop dates with no indefinite extensions.
2. Key Requirements and Obligations
The regulation creates two distinct compliance tracks — one for dental amalgam and one for mercury-containing lamps. Both carry manufacturing, import, export, and use restrictions with different phase-in dates. Understanding the full regulatory frameworks that govern restricted substances in the EU is essential for compliance planning.
Dental Amalgam Track:
✓ Use ban (general): No dental practitioner may use mercury amalgam on any patient except where a specific, documented medical need exists — effective January 1, 2025
✓ Export ban: No entity may export dental amalgam from the EU to any third country — effective January 1, 2025
✓ Derogation expiry: Member States that obtained temporary extensions based on healthcare reimbursement system needs must comply by June 30, 2026
✓ Manufacturing and import ban: No entity may manufacture or import dental amalgam into the EU — effective July 1, 2026
Mercury Lamp Track:
✓ Set 1 lamps: Manufacture and export of the first category of mercury-containing lamps banned — effective December 31, 2025
✓ Set 2 lamps: Manufacture and export of the final remaining lamp categories banned — effective December 31, 2026
Each obligation requires documented compliance evidence. The ability to stay audit-ready across frameworks ensures organizations can produce this evidence on demand during inspections or market surveillance actions.
3. Phased Compliance Deadlines: Full Timeline
The regulation uses a staggered enforcement approach. This creates urgency at each phase, not just at the final deadline. Operations leaders must map each deadline to specific product lines and supplier contracts.
Affected Category | Action Required | Deadline |
|---|---|---|
Dental Amalgam (Use) | General ban on use for all patients (exceptions only for specific medical needs) | Jan 1, 2025 |
Dental Amalgam (Export) | Prohibition on exporting dental amalgam to third countries | Jan 1, 2025 |
Mercury Lamps (Set 1) | Ban on manufacture/export of the first category of lamps | Dec 31, 2025 |
Dental Amalgam (Derogation) | Expiry of temporary extension for Member States with specific healthcare reimbursement needs | June 30, 2026 |
Dental Amalgam (Mfg/Import) | Prohibition on the manufacture and import of dental amalgam into the EU | July 1, 2026 |
Mercury Lamps (Set 2) | Ban on manufacture/export of the final remaining lamp categories | Dec 31, 2026 |
⚠ The first two deadlines have already passed. Organizations that have not yet confirmed compliance with the January 1, 2025 dental amalgam use and export bans are already in violation. For those approaching the December 2025 and 2026 lamp deadlines, the window for product redesign, supplier substitution, and inventory management is narrowing rapidly. Proactive compliance risk management must begin now.
4. Affected Industries and Products
The regulation's impact extends across four primary industry sectors. Companies operating in any of these areas within EU markets must evaluate their exposure immediately. Understanding market readiness vs. risk exposure is critical for executive decision-making.
Industry Sector | Specific Impact | Key Products Affected |
|---|---|---|
Healthcare & Dentistry | Dental practices, clinics, and dental material suppliers must eliminate amalgam use and sourcing | Dental amalgam capsules, mercury-based restorative materials |
Lighting & Electronics | Manufacturers, importers, and exporters of fluorescent and specialized mercury-added lamps face manufacturing and export bans | Compact fluorescent lamps, linear fluorescent tubes, high-intensity discharge lamps, specialty mercury lamps |
Waste Management | Entities handling hazardous mercury waste face expanded obligations for dental and electronic equipment disposal | Mercury-containing dental waste, end-of-life fluorescent lamps, mercury-contaminated equipment |
Chemical Manufacturing | Producers of mercury compounds and mercury-added products must cease production for banned applications | Mercury compounds, mercury-added product components |
For healthcare and dental organizations, the medical devices industry compliance requirements page provides context on how substance restrictions affect healthcare supply chains.
For lighting and electronics manufacturers, this regulation operates alongside the EU RoHS framework and the WEEE Directive, which already impose restrictions on mercury in electrical and electronic equipment. Organizations must coordinate compliance across all three frameworks. For a detailed breakdown of WEEE obligations, see the complete WEEE compliance guide for manufacturers.
For waste management entities, the expanded scope increases hazardous waste classification and specialized disposal requirements for both dental mercury waste and end-of-life mercury lamps. ESG and environmental data collection across the supply chain becomes more complex under these expanded obligations.
5. Dental Amalgam Ban: Detailed Obligations
The dental amalgam phase-out is the regulation's most sweeping provision. It eliminates a restorative material that has been in widespread use for over a century. Quality directors at dental supply companies must ensure product portfolios are fully transitioned.
What is now banned:
⚠ Use of dental amalgam on any patient (general population) — already in effect since Jan 1, 2025
⚠ Export of dental amalgam to any third country — already in effect since Jan 1, 2025
⚠ Manufacture and import of dental amalgam into the EU — effective July 1, 2026
What is temporarily permitted:
✓ Use of dental amalgam where a dental practitioner deems it strictly necessary for a specific patient based on documented medical need
✓ Member States that received temporary derogations based on healthcare reimbursement system needs may continue use until June 30, 2026 — not beyond
Compliance actions required:
Dental material suppliers must remove amalgam products from EU distribution
Manufacturers must cease amalgam production for EU markets by July 1, 2026
Exporters must confirm zero amalgam shipments to any destination since January 1, 2025
Dental clinics must document any medical-need exception use with auditable patient records
All entities must retain continuous audit-ready documentation proving compliance at each phase
The transition to composite resin alternatives requires supplier qualification, material testing, and clinical workflow updates. Launching new compliant products faster while maintaining quality standards is a critical operational priority for dental supply manufacturers.
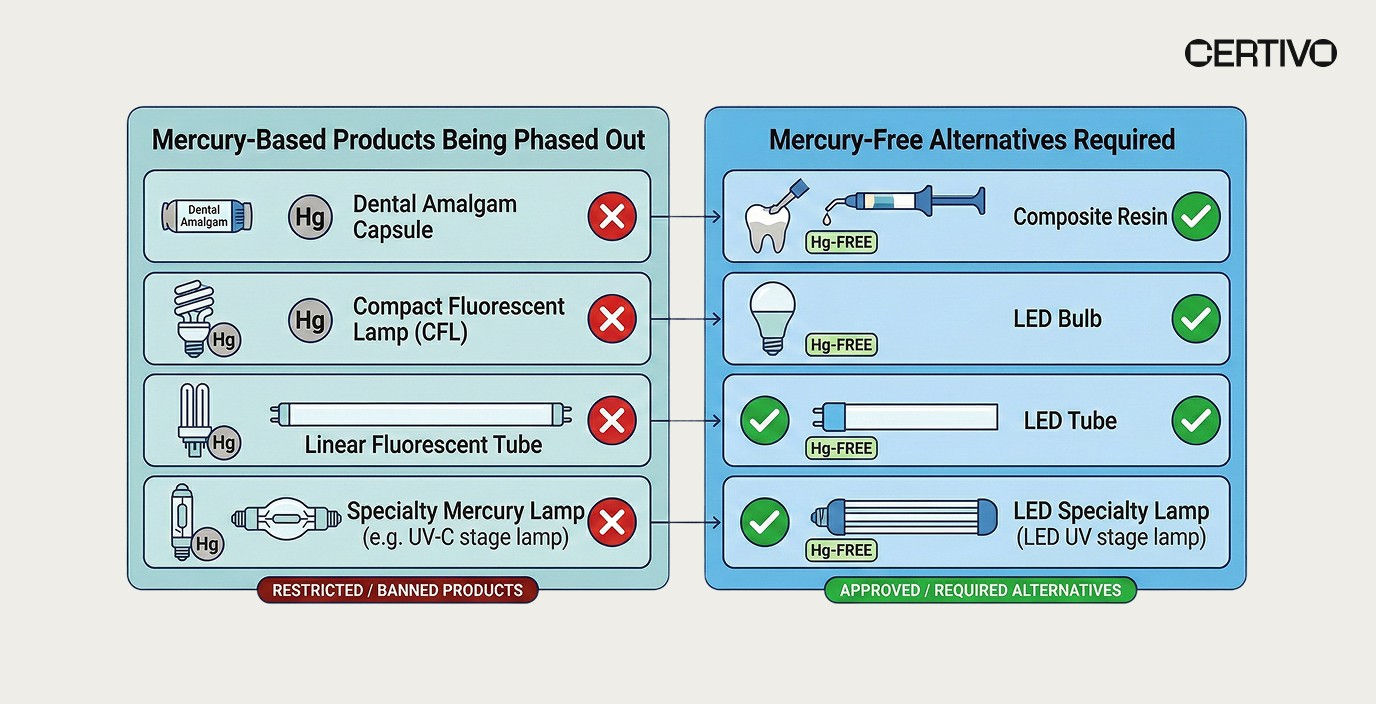
6. Mercury Lamp Ban: Detailed Obligations
The lamp provisions target six additional categories of mercury-containing lamps that were previously permitted under the original regulation. The ban covers both manufacture and export — meaning EU-based manufacturers cannot continue production even for non-EU markets. Industrial electronics manufacturers and lighting companies must evaluate their full product portfolios.
Set 1 lamps (banned by December 31, 2025):
⚠ First category of mercury-containing fluorescent and specialty lamps
⚠ Manufacture and export both prohibited
Set 2 lamps (banned by December 31, 2026):
⚠ Final remaining mercury lamp categories
⚠ Manufacture and export both prohibited
Compliance actions required:
Identify all mercury-containing lamp SKUs across manufacturing and export portfolios
Develop and qualify LED or other mercury-free replacements
Update product specifications, packaging, and technical documentation
Notify distributors and customers of product transitions and discontinuations
Ensure BOM-level material mapping captures mercury content at the component level for every lamp product
For manufacturers also managing RoHS compliance obligations, note that EU RoHS already restricts mercury in electrical and electronic equipment with specific exemptions for certain lamp types. The Mercury Regulation's ban now overrides several of those exemptions — manufacturers must cross-reference both frameworks. The recent EU RoHS lead exemption changes illustrate how substance-specific exemptions can shift rapidly.
Organizations exporting to markets beyond the EU should also assess whether mercury lamp restrictions create opportunities to expand into new markets faster with mercury-free product lines that meet emerging global standards.
7. Compliance Risks and Penalties
Non-compliance with the EU Mercury Regulation carries significant enforcement consequences. Member States are responsible for establishing penalties, which must be "effective, proportionate, and dissuasive" under EU law. Procurement and supply chain leaders must factor these risks into supplier qualification processes.
⚠ Market access loss: Products containing banned mercury substances cannot be legally placed on the EU market
⚠ Import seizure: Customs authorities can seize and destroy non-compliant shipments at EU borders
⚠ Financial penalties: Member State enforcement agencies impose fines that vary by jurisdiction but can be substantial
⚠ Product recalls: Non-compliant products already on the market may be subject to mandatory recall
⚠ Criminal liability: In certain Member States, knowing violations of banned substance regulations can trigger criminal prosecution
⚠ Reputational damage: Non-compliance with environmental health regulations carries significant brand and customer trust risk
The regulation's export ban provisions are particularly noteworthy. Unlike some EU chemical restrictions that apply only to products placed on the EU market, this regulation also prohibits manufacturing for export. EU-based manufacturers cannot shift production to serve non-EU markets — they must cease mercury lamp and dental amalgam production entirely. Understanding why compliance teams must drive strategic decisions is essential in this context.
8. Operational and Supply Chain Impact
The regulation creates cascading operational changes across procurement, manufacturing, product development, and distribution. For a broader perspective on managing documentation demands, review how tariffs and reshoring are increasing material compliance workloads.
For dental material suppliers and manufacturers:
✓ Reformulate product portfolios to eliminate mercury-based restorative materials
✓ Qualify composite resin alternatives with clinical validation data
✓ Update supplier contracts to prohibit mercury-containing inputs
✓ Manage inventory wind-down of existing amalgam stocks before manufacturing ban date
✓ Issue supplier compliance declarations confirming mercury-free material sourcing
For lighting manufacturers:
✓ Redesign or discontinue all mercury-containing lamp product lines
✓ Transition manufacturing processes to LED or equivalent mercury-free technologies
✓ Update product compliance documentation, CE marking, and technical files
✓ Coordinate with supplier self-service compliance portals to verify component-level mercury content
✓ Align product transitions with WEEE framework requirements for end-of-life obligations
For waste management entities:
✓ Update hazardous waste classification protocols for mercury-containing dental and lamp waste
✓ Ensure disposal facilities meet EU POPs and hazardous substance handling requirements
✓ Retain documentation of mercury waste volumes, treatment methods, and disposal destinations
Organizations managing compliance across multiple EU chemical regulations should consider centralizing market readiness from silos to a single source of truth to avoid duplicated effort and missed deadlines.
9. Compliance Readiness Checklist
📌 Use this checklist to assess your organization's preparedness for the EU Mercury Regulation amendments:
# | Action Item | Deadline Relevance | Status |
|---|---|---|---|
1 | Confirm dental amalgam use has ceased for general patients (medical-need exceptions documented) | Jan 1, 2025 ✅ | ☐ |
2 | Confirm zero dental amalgam exports since January 1, 2025 | Jan 1, 2025 ✅ | ☐ |
3 | Identify all mercury-containing lamp SKUs in manufacturing and export portfolios | Dec 31, 2025 | ☐ |
4 | Cease manufacture and export of Set 1 mercury lamps | Dec 31, 2025 | ☐ |
5 | Confirm LED or mercury-free alternatives are qualified and available for all affected lamp categories | Dec 31, 2025 / Dec 31, 2026 | ☐ |
6 | Verify Member State derogation status — confirm expiry compliance by June 30, 2026 | June 30, 2026 | ☐ |
7 | Cease all dental amalgam manufacturing and import into the EU | July 1, 2026 | ☐ |
8 | Cease manufacture and export of Set 2 mercury lamps | Dec 31, 2026 | ☐ |
9 | Update product technical documentation, CE files, and compliance declarations | All phases | ☐ |
10 | Retain audit-ready records for each phase of compliance | All phases | ☐ |
For organizations still managing compliance tracking through manual methods, building a future-ready compliance infrastructure provides a practical framework for transitioning to scalable systems. IT and systems leaders should be directly involved in evaluating platform readiness.
10. The Role of AI in Mercury Compliance Monitoring
The EU Mercury Regulation's phased enforcement structure — with multiple product categories, overlapping deadlines, and cross-framework interactions with RoHS and WEEE — creates complexity that exceeds what manual compliance processes can manage reliably. For a practical view of the difference AI makes, see a compliance engineer's week with and without AI.
Key challenges that benefit from AI-native compliance automation:
✓ BOM-level substance identification: Scanning product composition data to flag mercury content at the component level across all product lines
✓ Multi-framework cross-referencing: Automatically mapping mercury restrictions across EU Mercury Regulation, RoHS, WEEE, and POPs frameworks to identify overlapping obligations
✓ Supplier compliance verification: Aggregating supplier declarations and flagging gaps in mercury-free certifications
✓ Phased deadline tracking: Generating alerts for each compliance milestone across product categories
✓ Regulatory horizon scanning: Monitoring future amendments, implementing country-specific derogation tracking, and flagging emerging mercury restrictions globally
For organizations managing substance compliance across multiple regulatory frameworks, the complete guide to AI tools for compliance management details how automation addresses cross-framework complexity. Teams evaluating end-to-end product compliance tools should prioritize platforms that integrate substance-level tracking with regulatory intelligence.
The ability to standardize compliance across plants and regions is particularly important for multinational manufacturers who must coordinate mercury phase-out across EU facilities, export operations, and global supply chains simultaneously. AI in supply chain compliance management provides further context on how this works at scale.
11. Frequently Asked Questions (FAQs)
Q1: Is dental amalgam completely banned in the EU?
For general use, yes. Since January 1, 2025, dental practitioners may not use mercury amalgam on any patient unless a documented, specific medical need exists. From July 1, 2026, the manufacture and import of dental amalgam into the EU will also be prohibited. The medical devices industry compliance page provides additional context on how substance bans affect healthcare supply chains.
Q2: Can EU manufacturers still produce mercury lamps for export to non-EU countries?
No. The regulation bans both manufacture and export of affected mercury lamp categories. EU-based manufacturers cannot continue production for any market — domestic or international. Set 1 lamps must cease by December 31, 2025, and Set 2 lamps by December 31, 2026. This is a critical distinction from some other EU regulations. Understanding REACH and compliance lessons for 2026 provides context on how EU substance bans interact with export obligations.
Q3: Does the regulation affect Northern Ireland?
Yes. The regulation applies to Northern Ireland under the Windsor Framework. Manufacturers and importers operating in Northern Ireland must comply with all provisions. Expanding into new markets with compliance clarity requires understanding these jurisdictional details.
Q4: What are the mercury-free alternatives required for compliance?
For dental amalgam, the EU is driving adoption of composite resins and other mercury-free restorative materials. For lighting, LED technology is the primary replacement across all banned lamp categories. Organizations must qualify these alternatives through product testing and update technical documentation accordingly. Responding faster to customer RFQs with updated, compliant product data accelerates the transition.
Q5: How does this regulation interact with EU RoHS and WEEE?
EU RoHS already restricts mercury in electrical and electronic equipment but has granted specific exemptions for certain lamp types. The Mercury Regulation's lamp ban now overrides several of those RoHS exemptions. WEEE obligations for end-of-life lamp collection and recycling remain in effect. Organizations must cross-reference all three frameworks. The EU RoHS framework page and the WEEE compliance guide provide detailed breakdowns.
12. Conclusion
Regulation (EU) 2024/1849 eliminates the last intentional uses of mercury in the European Union. The dental amalgam ban and the mercury lamp phase-out create hard compliance deadlines that have already begun and extend through December 31, 2026. For manufacturers, importers, exporters, and healthcare suppliers, the EU mercury regulation 2026 compliance requirements are non-negotiable.
The phased timeline creates a false sense of distance. The first deadlines have already passed. The next — mercury lamp Set 1 manufacturing and export bans — arrives at the end of this year. Organizations that delay compliance planning risk market access loss, import seizures, financial penalties, and reputational damage.
📌 The official regulation text is available on the Official Journal of the EU. Additional policy context is available from the European Commission Mercury Policy page.
Whether your organization is phasing out dental amalgam, transitioning lamp manufacturing to LED, or managing mercury waste obligations, the compliance path requires documented evidence at every phase. To explore how AI-driven compliance platforms like Certivo support continuous regulatory readiness, connect with the Certivo team.
Lavanya
Lavanya is an accomplished Product Compliance Engineer with over four years of expertise in global environmental and regulatory frameworks, including REACH, RoHS, Proposition 65, POPs, TSCA, PFAS, CMRT, FMD, and IMDS. A graduate in Chemical Engineering from the KLE Institute, she combines strong technical knowledge with practical compliance management skills across diverse and complex product portfolios.
She has extensive experience in product compliance engineering, ensuring that materials, components, and finished goods consistently meet evolving international regulatory requirements. Her expertise spans BOM analysis, material risk assessments, supplier declaration management, and test report validation to guarantee conformity. Lavanya also plays a key role in design-for-compliance initiatives, guiding engineering teams on regulatory considerations early in the product lifecycle to reduce risks and streamline market access.
Her contributions further extend to compliance documentation, certification readiness, and preparation of customer deliverables, ensuring transparency and accuracy for global stakeholders. She is adept at leveraging compliance tools and databases to efficiently track regulatory changes and implement proactive risk mitigation strategies.
Recognized for her attention to detail, regulatory foresight, and collaborative approach, Lavanya contributes significantly to maintaining product compliance, safeguarding brand integrity, and advancing sustainability goals within dynamic, globally integrated manufacturing environments.