
Lavanya

If your organization places chemical mixtures on the European market, a major regulatory shift is about to redefine how you classify, label, and document your products. The European Chemicals Agency (ECHA) has introduced three new hazard classes under the EU CLP Regulation — effective May 1, 2026 — and the compliance impact reaches far deeper than a labelling update.
Manufacturers, formulators, and importers must now classify mixtures for Endocrine Disruptors (ED), Persistent, Bioaccumulative and Toxic substances (PBT/vPvB), and Persistent, Mobile and Toxic substances (PMT/vPvM). These EU CLP new hazard classes also trigger mandatory Safety Data Sheet (SDS) updates across four critical sections. Miss the deadline and your products face EU market withdrawal.
The question every compliance director and CEO should be asking right now: Do we know which of our mixtures contain substances classified under the new hazard categories — and can we prove it?
If the answer is uncertain, your organization is already behind. Chemical and hazmat compliance teams must begin classification assessments immediately. This article gives you the complete regulatory breakdown, operational impact analysis, and step-by-step compliance roadmap.
📊 Not sure how many of your products are affected? Most manufacturers we work with discover 30–60% of their EU mixture portfolio requires reclassification under the new CLP hazard classes. Take Certivo's free Rapid Risk Assessment → Map your exposure in 60 seconds
📌 Table of Contents
What Changed: New Hazard Classes Under EU CLP
The Three New Hazard Classes Explained
Safety Data Sheet (SDS) Update Requirements
Timeline: Effective Dates and Transition Period
Industries and Products Affected
Compliance Risks and Penalties for Non-Classification
Operational and Supply Chain Impact
How to Classify Mixtures Under New CLP Hazard Classes — Step by Step
The Role of AI in CLP Compliance Monitoring
FAQs
Executive Conclusion
What Changed: Why the EU CLP Regulation Now Covers Entirely New Hazard Categories
The CLP Regulation (EC) No 1272/2008 has been the EU's backbone for classifying, labelling, and packaging chemical substances and mixtures for over 15 years. Until now, classifications focused on established hazard categories — flammability, acute toxicity, carcinogenicity, and environmental hazards.
That framework is no longer sufficient. Scientific evidence has established that certain chemical properties — endocrine disruption, environmental persistence combined with bioaccumulation, and environmental mobility — pose risks that previous CLP categories do not capture.
ECHA's amendment introduces three entirely new hazard classes that manufacturers must apply when classifying mixtures:
🧬 Endocrine Disruptors (ED) — substances that interfere with hormonal systems in humans or the environment
☣️ PBT / vPvB — Persistent, Bioaccumulative, and Toxic (or very Persistent and very Bioaccumulative) substances
💧 PMT / vPvM — Persistent, Mobile, and Toxic (or very Persistent and very Mobile) substances
The critical shift: These classes previously applied only to individual substances under the REACH framework. Now they extend to mixtures under CLP. Every formulated product containing ingredients with these properties must be evaluated, classified, and labelled under the new scheme. Organizations managing multi-framework obligations should understand how these changes intersect with existing REACH requirements.
⚠ This is not optional. Companies placing mixtures on the EU market after May 1, 2026 must comply. Mixtures already on the market receive a transitional period until May 1, 2028 — but the reclassification workload is substantial and waiting creates a dangerous bottleneck.
The real challenge isn't understanding the regulation. It's knowing whether your products contain the affected substances — across hundreds or thousands of formulations, sourced from dozens of global suppliers, many of whom may not yet have updated their own substance data. This is precisely why organizations that lack materials and environmental compliance infrastructure are most at risk.

The Three New Hazard Classes Explained: What Each Means for Your Products
Understanding what each hazard class requires is essential before starting classification. But the real question compliance teams face isn't "what are these classes" — it's "how do I identify which of my mixture ingredients fall into these categories across my entire product portfolio?"
Endocrine Disruptors (ED) — The Hardest to Track
Endocrine disruptors alter the functioning of the hormonal system in humans or wildlife. They cause adverse developmental, reproductive, neurological, and immune effects — often at very low concentrations, which makes them difficult to identify through standard testing.
📌 Why this is operationally difficult:
Many common ingredients in consumer products, cosmetics, and industrial formulations have ED properties that suppliers may not yet have disclosed
Classification requires substance-level data that many manufacturers do not currently hold for every mixture component
Organizations without BOM-level substance tracking will struggle to identify ED-classified ingredients across product portfolios
PBT / vPvB — Alignment with REACH, But New Mixture Obligations
PBT and vPvB substances persist in the environment, accumulate in living organisms, and cause toxic effects over time. These properties have long been tracked under REACH SVHC identification.
📌 What changes for mixtures:
Mixtures must now be evaluated for PBT/vPvB ingredient content — not just individual substances
Classification requires updated environmental and health hazard labelling on all affected product packaging
This aligns with REACH SVHC and Annex XVII obligations but extends the labelling requirement to the mixture level
PMT / vPvM — A Completely New Classification Category
PMT and vPvM is the most novel of the three classes. These substances are persistent and highly mobile — spreading easily through water systems and soil. This class addresses growing regulatory concern about groundwater contamination.
📌 Why this matters for manufacturers:
There is no direct precedent for PMT/vPvM classification in previous CLP or REACH schemes
Supplier data on mobility properties is often incomplete or unavailable
Organizations will need to actively request new data from suppliers — many of whom may not yet be prepared to provide it
Hazard Class | Description | Primary Concern | Compliance Challenge |
|---|---|---|---|
ED | Endocrine Disruptors | Hormonal system interference | Low-concentration identification across complex formulations |
PBT / vPvB | Persistent, Bioaccumulative, Toxic | Long-term bioaccumulation | Aligning REACH substance data with CLP mixture classification |
PMT / vPvM | Persistent, Mobile, Toxic | Water contamination, mobility | Novel category — limited supplier data availability |
Organizations managing PFAS-containing products should note significant overlap — many PFAS compounds meet PBT and PMT criteria. For context on parallel PFAS regulations, see the global PFAS compliance master guide.
Safety Data Sheet (SDS) Update Requirements: The Hidden Workload
The new hazard classes don't just affect product labels — they trigger mandatory revisions to Safety Data Sheets across four specific sections. For organizations managing hundreds or thousands of mixture SDS, this is where the operational burden becomes most acute.
Compliance regulation managers must activate SDS revision workflows now — not when the deadline is imminent.
📌 SDS sections requiring updates:
SDS Section | Content Area | What Must Change | Operational Impact |
|---|---|---|---|
Section 2 | Hazard Identification | Add new hazard class statements for ED, PBT/vPvB, PMT/vPvM | Every affected product SDS must be revised |
Section 3 | Composition / Ingredients | Identify ingredients classified under new hazard classes | Requires verified substance-level classification data from suppliers |
Section 11 | Toxicological Information | Include endocrine disruption data and PBT/vPvB assessment | May require new data collection from substance manufacturers |
Section 12 | Ecological Information | Include PMT/vPvM environmental fate and mobility data | Novel data requirement — many suppliers lack this information |
⚠ The scale problem: A mid-size chemical manufacturer with 500 mixture products may need to revise 200–300 SDS documents. Each revision requires verified ingredient classification data, updated hazard statements, and audit-ready documentation. Doing this manually — with spreadsheets, emails, and PDF reviews — is not feasible within the available timeline.
This is precisely where organizations lose months. The data exists across supplier SDS, technical datasheets, REACH registration dossiers, and internal formulation records — but it is fragmented. Automated document parsing and certificate validation can extract, validate, and propagate substance data across all affected SDS in a fraction of the time.
⚠ Facing hundreds of SDS revisions before May 2026? This is the #1 compliance bottleneck manufacturers tell us about. Certivo's AI-powered document engine automates bulk SDS updates — screening formulations, flagging affected mixtures, and generating audit-ready SDS across your entire portfolio. See how Certivo automates SDS compliance at scale →
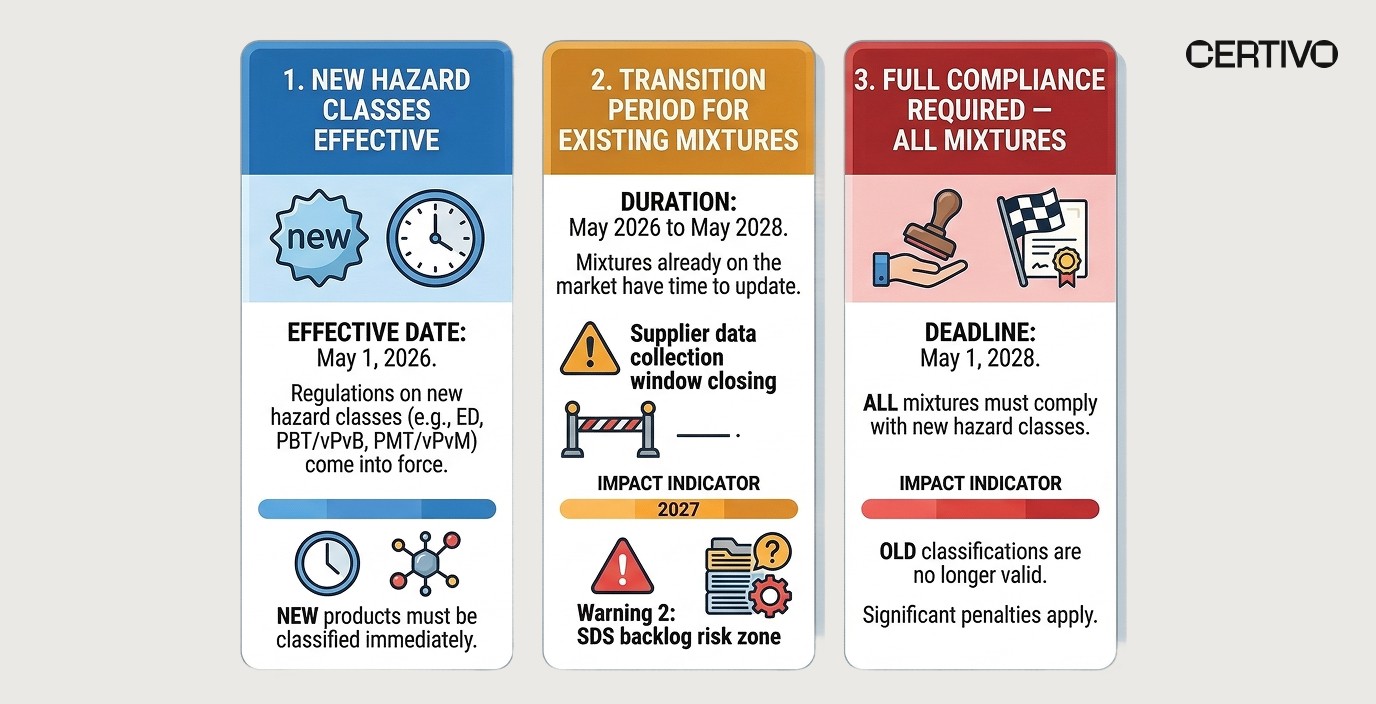
Timeline: Effective Dates, Transition Period, and Why Waiting Until 2028 Is a Trap
The regulation establishes a two-phase timeline. But the transition period is not the safety net most compliance teams think it is. VP and Director-level quality leaders should map these milestones against internal product launch calendars immediately.
Milestone | Date | What It Means |
|---|---|---|
New hazard classes effective for mixtures | May 1, 2026 | All new mixtures placed on EU market must comply from day one |
Transition period for existing mixtures | Until May 1, 2028 | Existing products may continue with previous classification |
Full compliance — no exceptions | May 1, 2028 | Every mixture on the EU market must reflect new hazard classifications |
📊 Why waiting until 2028 is dangerous:
Supplier data collection takes 6–12 months for complex supply chains — if you start in 2027, you will not finish in time
SDS revision backlogs compound exponentially — companies that delay will compete with every other manufacturer for the same limited regulatory consulting resources
Customer requirements will accelerate ahead of the legal deadline — major EU distributors and OEMs will demand compliant SDS well before May 2028
New product launches after May 2026 cannot use the transition period at all — they must comply from market placement
Organizations still running classification workflows through spreadsheets should urgently evaluate replacing manual systems with scalable compliance infrastructure before the timeline compresses further.
Industries and Products Affected: Is Your Sector Exposed?
The new CLP hazard classes affect any organization that formulates, manufactures, imports, or distributes chemical mixtures within the EU. But the impact severity varies significantly by sector. For a full view of product compliance management across industries, organizations should assess their complete product portfolio.
Industry | Affected Products | Impact Level | Primary Challenge |
|---|---|---|---|
Chemical Manufacturing | Formulated substances, intermediates, processing aids | ⚠ Critical | Thousands of formulations requiring reclassification |
Paints, Coatings & Adhesives | Industrial coatings, sealants, adhesive formulations | ⚠ High | ED and PBT substances common in raw materials |
Consumer Products | Cleaning agents, detergents, household chemicals | ⚠ High | Supply chain transparency gaps |
Cosmetics & Personal Care | Formulated cosmetic mixtures, fragrances | ⚠ High | ED substances prevalent in fragrance and preservative ingredients |
Pharmaceuticals & Laboratories | Chemical preparations, lab reagents, excipients | ⚠ Moderate–High | Novel PMT data requirements |
Industrial Manufacturing | Lubricants, metalworking fluids, process chemicals | ⚠ High | Multi-tier supply chain data gaps |
Chemical manufacturers face the most intensive compliance burden. They formulate and supply the mixtures that downstream industries use — meaning upstream classification errors cascade through the entire value chain.
For consumer goods companies, products containing fragrances, surfactants, or preservatives with endocrine-disrupting properties will require reclassification and relabelling. Brands should also monitor how this intersects with the global cosmetics PFAS and microplastics regulatory shifts.
Pharmaceutical and biotech organizations handling chemical preparations and laboratory reagents must evaluate whether any mixture components trigger the new hazard classifications.
🏭 How exposed is your product portfolio? Certivo's BOM-level substance screening identifies every ED, PBT, and PMT classified ingredient across your entire mixture portfolio — giving you a clear compliance gap report before you commit resources. Book a free compliance exposure assessment →
Compliance Risks and Penalties for Non-Classification
This is not a soft regulation with advisory timelines. CLP enforcement is managed by national authorities across EU Member States, and non-compliance carries material consequences. Organizations must manage compliance risk proactively — not reactively.
What happens if you don't comply:
Risk | Impact | Who Bears It |
|---|---|---|
Regulatory fines | Substantial financial penalties varying by Member State | Manufacturer / Importer |
EU market withdrawal | Non-compliant mixtures ordered off shelves and out of distribution | Manufacturer + Distributor |
Supply chain rejection | Downstream customers refuse non-compliant SDS; contract termination | Supplier / Formulator |
Customer liability transfer | Distributors and OEMs held liable for placing non-compliant products | Entire value chain |
Reputational damage | Loss of B2B trust; negative audit outcomes; customer churn | Brand / Organization |
⚠ The risk most organizations underestimate: Even if your products are correctly classified, if your SDS is not updated, you are non-compliant. SDS is the legally binding communication mechanism — an outdated SDS with missing hazard classifications is a regulatory violation regardless of whether the product itself is safe.
The transition period to May 2028 gives existing products a grace window, but it does not protect organizations that make no effort to reclassify. Enforcement authorities distinguish between companies in active transition and companies that have done nothing. For a broader perspective on scaling compliance, see why people-only compliance can't scale.
Operational and Supply Chain Impact: Where Compliance Actually Breaks Down
The regulation is clear. The challenge is execution. Most compliance failures under CLP are not caused by misunderstanding the law — they are caused by data gaps, supplier non-response, fragmented systems, and manual workflows that cannot scale. Procurement and supply chain leaders must be involved from day one.
The Supplier Data Problem
Your classification is only as good as the data your suppliers provide. Under the new CLP hazard classes:
⚠ Suppliers must provide updated classification data for every substance that may meet ED, PBT/vPvB, or PMT/vPvM criteria
⚠ Many suppliers — particularly smaller or non-EU based suppliers — do not yet have this data available
⚠ Chasing individual suppliers via email for substance classification data is the single biggest time drain in CLP compliance
Streamlining supplier documentation through structured collection portals is the operational difference between on-time compliance and missed deadlines.
The Cross-Functional Coordination Challenge
CLP reclassification is not a regulatory affairs task alone. It requires coordinated action across:
R&D — formulation review and ingredient substitution assessment
Procurement — supplier data requests and alternative sourcing
Quality — SDS revision and label redesign approval
Operations — production label changes and inventory management
Sales — downstream customer communication and compliant documentation distribution
Organizations managing products across multiple EU member states should standardize compliance across plants and regions to prevent inconsistent classification outcomes.
For companies simultaneously managing REACH, RoHS, PFAS, and CLP obligations, the ability to maintain a centralized compliance data backbone eliminates duplication and ensures classification decisions are consistent across all frameworks.
🔗 Struggling with supplier data gaps for the new CLP hazard classes? Certivo's automated supplier portals send structured data requests, track response rates, validate incoming SDS and certificates, and flag non-responsive suppliers — eliminating months of manual follow-up. See how Certivo automates supplier compliance data collection →
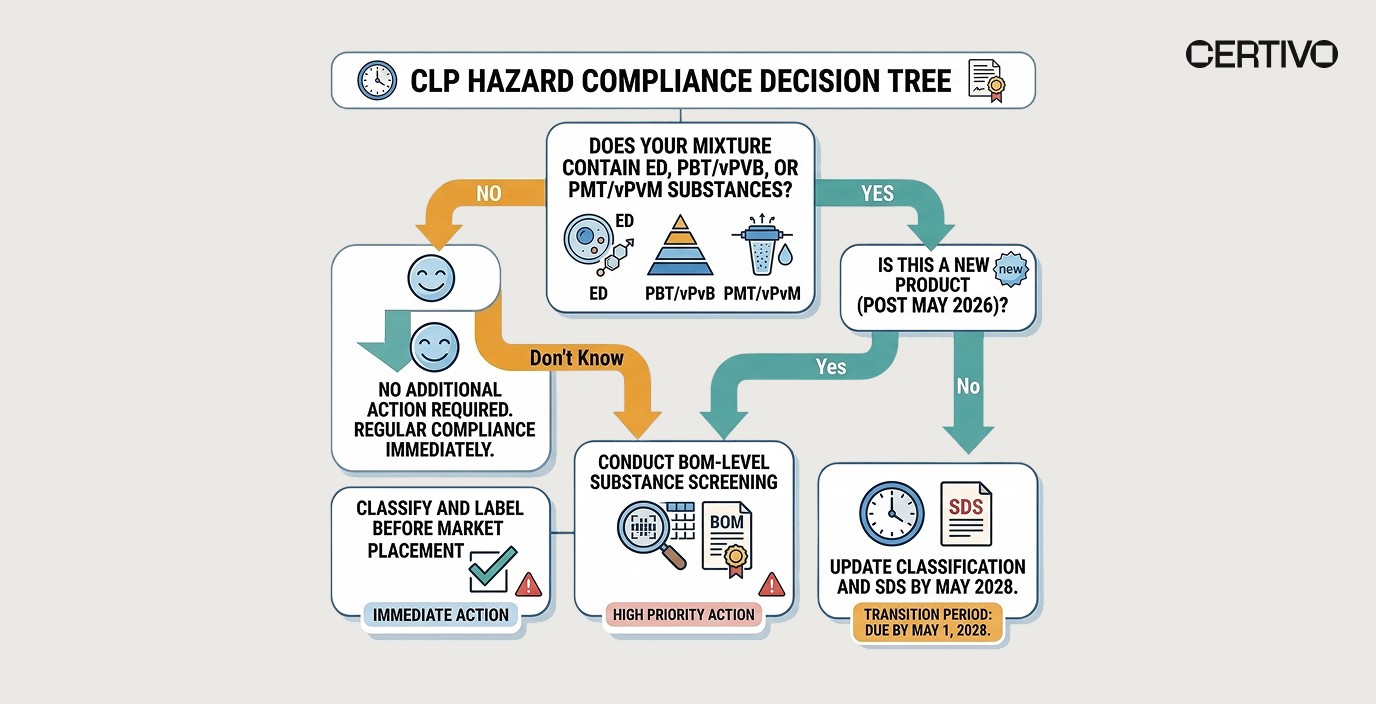
How to Classify Mixtures Under New CLP Hazard Classes — Step by Step
📌 This is the actionable compliance roadmap your team needs:
Phase 1: Assessment (Start Immediately)
Step | Action | Owner | Certivo Capability |
|---|---|---|---|
1 | Inventory all mixtures currently placed on the EU market | Regulatory Affairs | |
2 | Identify all mixture ingredients that may be classified as ED, PBT/vPvB, or PMT/vPvM | R&D + Compliance | BOM-level substance screening |
3 | Request updated classification data from all upstream suppliers | Procurement |
Phase 2: Classification & Documentation (Q3 2025 – Q1 2026)
Step | Action | Owner | Certivo Capability |
|---|---|---|---|
4 | Reclassify all affected mixtures under the new hazard categories | Regulatory Affairs | Automated hazard class mapping |
5 | Update SDS Sections 2, 3, 11, and 12 for every affected product | EHS + Regulatory | AI-powered SDS revision engine |
6 | Redesign product labels with new hazard statements and pictograms | Packaging + Regulatory | Label compliance workflows |
Phase 3: Communication & Monitoring (Q2 2026 Onward)
Step | Action | Owner | Certivo Capability |
|---|---|---|---|
7 | Distribute updated SDS and labels to downstream customers | Sales + Compliance | |
8 | Train relevant staff on new hazard class criteria | EHS + HR | Compliance documentation access |
9 | Establish continuous monitoring for ECHA guidance updates | Regulatory Affairs | Regulatory horizon scanning |
10 | Maintain audit-ready documentation for all classification decisions | Compliance |
CEOs and founders should ensure executive sponsorship for this roadmap — CLP reclassification is a market access requirement, not a back-office task.
For teams evaluating whether current systems can handle this workload, this guide to building a future-ready compliance infrastructure provides a practical assessment framework.
📋 Want this compliance roadmap customized for your product portfolio? Certivo's compliance team will map your specific mixture portfolio against the new CLP hazard classes and deliver a prioritized action plan — so you know exactly what needs to change, by when, and how. Request your custom CLP compliance roadmap →
The Role of AI in CLP Hazard Class Compliance: Why Manual Processes Won't Scale
The scale of this CLP update — potentially affecting thousands of mixtures across complex product portfolios — exceeds what manual compliance workflows can deliver within the available timeline. This is not an efficiency preference. It is a capacity reality. For a practical comparison of manual vs. AI-powered compliance workflows, see a compliance engineer's week with and without AI.
The manual approach timeline for a 500-product portfolio:
Task | Manual Time | AI-Automated Time |
|---|---|---|
Ingredient screening for ED/PBT/PMT across all formulations | 8–12 weeks | 2–3 days |
Supplier data collection and validation | 6–12 months | 4–6 weeks |
SDS revision (Sections 2, 3, 11, 12) for affected products | 4–8 months | 2–4 weeks |
Audit-ready documentation generation | Ongoing manual effort | Automatic |
What AI-native compliance platforms deliver:
✓ Automated BOM and formulation scanning — flags every ingredient classified as ED, PBT/vPvB, or PMT/vPvM across your entire portfolio in days, not months
✓ Supplier data portals — structured questionnaires sent automatically, with response tracking, validation, and escalation for non-responsive suppliers
✓ Bulk SDS revision — propagates classification changes across all affected product documents simultaneously
✓ Regulatory horizon scanning — alerts when ECHA publishes new guidance, updates classification criteria, or issues enforcement notices
✓ Document parsing — extracts substance data from supplier SDS, certificates, and material declarations automatically
For a comprehensive overview, see AI tools for compliance management: the complete guide. IT and systems leaders should evaluate how compliance platforms integrate with existing ERP and PLM systems to ensure classification data flows consistently.
🤖 See what AI-powered CLP compliance looks like in practice Watch a live walkthrough of how Certivo automates substance screening, SDS updates, and supplier data collection — from a single platform. Watch the on-demand product demo →
Frequently Asked Questions (FAQs)
Q1: What are the new EU CLP hazard classes for mixtures and how do they affect my products?
The three new hazard classes are Endocrine Disruptors (ED), Persistent, Bioaccumulative and Toxic (PBT/vPvB), and Persistent, Mobile and Toxic (PMT/vPvM). They affect any mixture placed on the EU market that contains ingredients meeting these classification criteria. This means every formulated product — from industrial coatings to consumer detergents — must be screened. For context on how CLP and REACH obligations overlap, review the REACH framework overview.
Q2: When do the new CLP hazard classes take effect and how much time do I have?
New mixtures placed on the EU market after May 1, 2026 must comply immediately. Existing products receive a transition until May 1, 2028. However, supplier data collection and SDS revision timelines mean organizations should begin now — starting in 2027 is likely too late for complex portfolios.
Q3: Which SDS sections must be updated and what data do I need from suppliers?
Companies must update Section 2 (Hazard Identification), Section 3 (Composition), Section 11 (Toxicological Information), and Section 12 (Ecological Information). You need verified substance classification data from every supplier whose ingredients may meet ED, PBT, or PMT criteria. Automated supplier data collection dramatically reduces the time and effort required.
Q4: How do I know if my mixture ingredients are classified as ED, PBT, or PMT?
Check the ECHA Classification & Labelling Inventory and your suppliers' substance registration dossiers. For large portfolios, manual ingredient-by-ingredient checking is impractical. AI-powered BOM screening tools can scan your full formulation database and flag affected ingredients automatically. Tracking compliance at the BOM level is essential for this process.
Q5: What happens if I don't reclassify my mixtures by the deadline?
Non-compliant mixtures face EU market withdrawal, regulatory fines (varying by Member State), supply chain rejection from downstream customers, and potential liability for distributors and OEMs. Even correctly formulated products are non-compliant if their SDS is not updated. Electronics manufacturers using process chemicals should also evaluate their exposure.
Conclusion: The Window to Act Is Now
The introduction of EU CLP new hazard classes for mixtures — Endocrine Disruptors, PBT/vPvB, and PMT/vPvM — is not a minor regulatory update. It is a structural expansion of how every chemical mixture sold in Europe must be classified, labelled, and documented. The compliance impact touches product formulation, SDS documentation, supplier data management, product labelling, customer communication, and market access strategy.
The facts are clear:
✓ May 1, 2026: new products must comply
✓ May 1, 2028: all products must comply
⚠ Supplier data collection takes 6–12 months
⚠ SDS revision at portfolio scale takes 4–8 months manually
⚠ Starting in 2027 means missing the deadline
Organizations that begin classification assessments, supplier engagement, and SDS revisions now will maintain uninterrupted EU market access. Those that delay will face bottlenecks, market withdrawal risk, and competitive disadvantage.
📌 For the official ECHA CLP guidance and hazard classification resources, visit ECHA's CLP information portal.
🚀 Your next step — choose the path that fits your stage:
Just learning? → Watch the on-demand AI compliance webinar
Need to assess exposure? → Take the free Rapid Risk Assessment (60 seconds)
Ready to act? → Talk to a Certivo compliance expert
Lavanya
Lavanya is an accomplished Product Compliance Engineer with over four years of expertise in global environmental and regulatory frameworks, including REACH, RoHS, Proposition 65, POPs, TSCA, PFAS, CMRT, FMD, and IMDS. A graduate in Chemical Engineering from the KLE Institute, she combines strong technical knowledge with practical compliance management skills across diverse and complex product portfolios.
She has extensive experience in product compliance engineering, ensuring that materials, components, and finished goods consistently meet evolving international regulatory requirements. Her expertise spans BOM analysis, material risk assessments, supplier declaration management, and test report validation to guarantee conformity. Lavanya also plays a key role in design-for-compliance initiatives, guiding engineering teams on regulatory considerations early in the product lifecycle to reduce risks and streamline market access.
Her contributions further extend to compliance documentation, certification readiness, and preparation of customer deliverables, ensuring transparency and accuracy for global stakeholders. She is adept at leveraging compliance tools and databases to efficiently track regulatory changes and implement proactive risk mitigation strategies.
Recognized for her attention to detail, regulatory foresight, and collaborative approach, Lavanya contributes significantly to maintaining product compliance, safeguarding brand integrity, and advancing sustainability goals within dynamic, globally integrated manufacturing environments.