
Kunal Chopra
A certificate of conformance (CoC) is a formal document issued by manufacturers or suppliers declaring that products, materials, or services meet specified requirements, standards, or contractual obligations. Unlike test reports that provide laboratory results, a CoC represents the supplier's attestation that delivered goods conform to agreed-upon specifications.
Certificate of conformance documentation has become central to supply chain compliance management as regulatory frameworks expand and customers demand verified conformity across REACH, RoHS, PFAS, and industry-specific standards. Understanding CoC requirements, proper documentation formats, and automation strategies determines operational efficiency and audit readiness.
Table of Contents
Certificate of Conformance Definition and Purpose
CoC vs Certificate of Analysis vs Certificate of Compliance
When a Certificate of Conformance is Required
Essential Elements of a Valid CoC
Industries Requiring Certificate of Conformance Documentation
Regulatory Frameworks Governing CoCs
Common Certificate of Conformance Template Requirements
Verification and Validation Challenges
Risks of Inadequate CoC Documentation
How to Manage CoC Documentation at Scale
AI-Native Automation for Certificate Management
Frequently Asked Questions
Conclusion
Certificate of Conformance Definition and Purpose
A certificate of conformance serves as a supplier's formal declaration that delivered products conform to specifications defined in purchase orders, technical drawings, industry standards, or regulatory requirements. The CoC certificate represents the supplier's legal attestation of conformity rather than independent verification.
Primary Functions of CoC Documentation
Contractual Compliance Verification: CoCs confirm that suppliers have delivered products matching purchase order specifications, material grades, dimensional tolerances, and performance characteristics. This documentation establishes accountability when products fail to meet agreed standards.
Regulatory Requirement Fulfillment: Many regulatory frameworks require manufacturers to maintain documented evidence of conformity. Medical device compliance, aerospace supply chain requirements, and automotive quality standards mandate CoC documentation for traceability and audit purposes.
Quality Assurance Documentation: CoCs form part of the quality record trail demonstrating that incoming materials meet specifications before entering production. This documentation supports root cause analysis when product failures occur.
Supply Chain Risk Management: Certificate of conformance requirements establish accountability across multi-tier supply chains. When subcontractors provide materials, CoCs create documentation trails linking finished products to source materials.
Audit Trail Creation: Regulatory audits, customer assessments, and internal quality reviews require documented evidence of supplier conformance. Continuous audit-ready documentation depends on systematic CoC collection and organization.
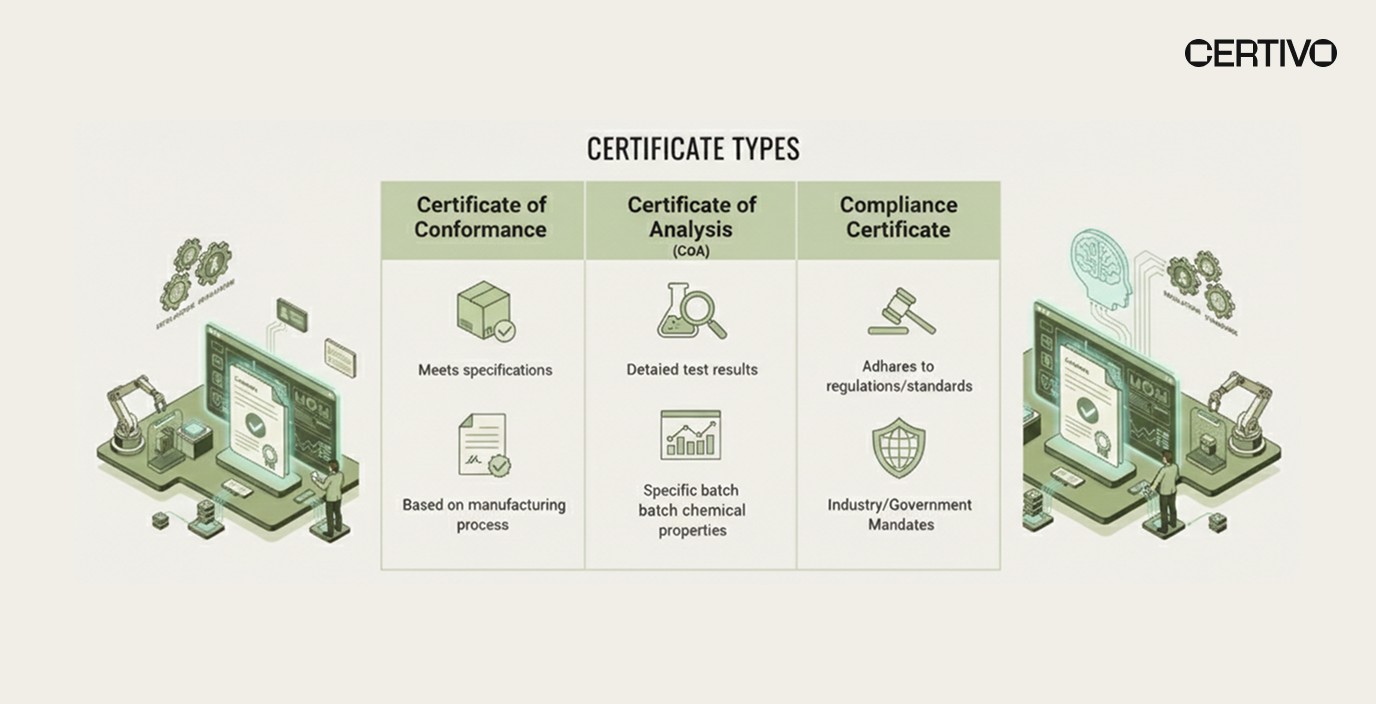
CoC vs Certificate of Analysis vs Certificate of Compliance
Confusion between certificates of conformance, certificates of analysis (CoA), and certificates of compliance stems from overlapping documentation requirements. Understanding the distinctions prevents documentation gaps during audits.
Key Document Distinctions
Document Type | Primary Purpose | Issued By | Content Focus |
|---|---|---|---|
Certificate of Conformance (CoC) | Attest conformity to specifications | Manufacturer/Supplier | Specification compliance declaration |
Certificate of Analysis (CoA) | Report test results and measurements | Testing laboratory or manufacturer | Quantitative test data and results |
Certificate of Compliance | Attest regulatory compliance | Manufacturer/Importer | Regulatory requirement conformity |
Certificate of Conformance: Declares that products meet specified requirements without necessarily providing detailed test data. May reference test reports but focuses on attestation rather than results. Common in manufacturing, construction, and industrial supply chains.
Certificate of Analysis: Provides detailed test results, measurements, and analytical data. Includes methodology, equipment used, detection limits, and quantitative findings. Common in pharmaceuticals, chemicals, and materials where composition verification is critical.
Certificate of Compliance: Specifically addresses regulatory compliance rather than general specifications. Often references specific regulations like RoHS compliance, REACH conformity, or industry standards. May be required for customs clearance or market access.
When Multiple Documents Are Required
Many transactions require both CoCs and CoAs. For example, semiconductor manufacturers may need supplier CoCs attesting to specification conformity plus CoAs providing composition analysis for restricted substances. Medical device companies require CoCs for component conformity plus certificates of compliance for regulatory frameworks.
When a Certificate of Conformance is Required
Certificate of conformance requirements vary by industry, product category, contractual terms, and regulatory mandates. Organizations must identify which transactions require CoCs and establish systematic collection processes.
Common CoC Requirement Triggers
Contractual Obligations: Purchase orders, supplier agreements, and master service agreements often specify CoC requirements. Contract language may require CoCs for specific materials, dimensional specifications, or performance characteristics.
Industry Standards Compliance: Standards bodies like ISO, ASTM, ASME, and ANSI establish CoC requirements for materials and components. Industrial automation and industrial electronics sectors frequently mandate CoCs for components.
Regulatory Framework Requirements: Frameworks like EU Batteries Regulation, WEEE, and Extended Producer Responsibility programs require documented evidence of conformity. Material restrictions under REACH and RoHS often necessitate supplier CoCs.
Customer Quality Requirements: OEMs and large buyers impose CoC requirements on suppliers as part of quality assurance programs. Aerospace manufacturers and automotive supply chains maintain strict CoC documentation requirements.
High-Risk Materials and Components: Critical safety components, pressure vessels, lifting equipment, and structural materials typically require CoCs regardless of contractual language. Liability considerations drive CoC requirements for high-consequence failure scenarios.
Import and Export Requirements: Customs authorities may require CoCs for specific product categories. Buy America Act compliance and origin verification often necessitate manufacturer CoCs.
Essential Elements of a Valid CoC
A valid certificate of conformance must contain specific information elements to serve its documentation purpose. Incomplete or ambiguous CoCs create compliance gaps during audits and investigations.
Mandatory CoC Information Elements
Supplier Identification: Full legal name, address, and contact information of the issuing organization. Ambiguous identification undermines accountability when conformity issues arise.
Customer Identification: Name and address of the purchasing organization. This links the CoC to specific transactions and prevents misapplication to unrelated shipments.
Purchase Order Reference: Specific purchase order number, line items, and revision levels. CoCs must clearly identify which contractual requirements they address.
Product Description: Detailed description including part numbers, revision levels, material grades, and quantities. Generic descriptions create ambiguity about what the CoC covers.
Specification References: Explicit citation of specifications, standards, drawings, or contractual requirements the product conforms to. References should include document numbers and revision levels.
Declaration Statement: Clear statement that the described products conform to referenced specifications. Ambiguous language like "substantially conforms" or "generally meets" undermines the declaration's value.
Date and Signature: Date of issuance and authorized signature. Electronic signatures are acceptable if they meet applicable digital signature standards.
Test Report References (if applicable): When conformity relies on test results, CoCs should reference specific test reports by number and date. This links declarations to supporting evidence.
Exceptions and Deviations: Any deviations from specifications must be explicitly noted. Implied conformity when known exceptions exist constitutes misrepresentation.
Optional But Valuable Information
Many organizations include additional information like batch numbers, manufacturing dates, expiration dates (for materials with shelf life), inspection records, and traceability codes. Streamlining supplier documentation benefits from standardized CoC templates specifying required and optional elements.
Industries Requiring Certificate of Conformance Documentation
CoC requirements span multiple industries with varying documentation standards and regulatory drivers. Understanding industry-specific requirements prevents compliance gaps.
High-CoC-Requirement Industries
Aerospace and Defense: Aerospace compliance demands rigorous CoC documentation for materials, components, and assemblies. AS9100 quality management systems mandate documented evidence of conformity. Material traceability requirements extend to raw material sources.
Medical Devices: Medical device manufacturers require CoCs for components as part of FDA Quality System Regulation (QSR) and ISO 13485 compliance. Biocompatibility requirements necessitate material conformity documentation.
Automotive: Automotive OEMs impose CoC requirements through IATF 16949 and customer-specific quality requirements. Automotive supply chains require documented conformity for safety-critical components.
Construction and Building Materials: Construction materials like steel, concrete, and structural components require CoCs certifying conformity to building codes and standards. Material test reports often accompany CoCs.
Semiconductor and PCB Manufacturing: Semiconductor manufacturers require CoCs for materials, chemicals, and components. Purity specifications and contamination control drive strict documentation requirements.
Energy Storage and Batteries: Battery manufacturers require CoCs for cells, separators, electrolytes, and other components. Safety testing and regulatory compliance necessitate comprehensive documentation.
Industrial Electronics: Industrial electronics manufacturers require CoCs for components, materials, and subassemblies. RoHS compliance and REACH conformity documentation often takes CoC form.
Test and Measurement Equipment: Test and measurement manufacturers require CoCs for precision components where calibration and tolerances are critical to equipment performance.
Regulatory Frameworks Governing CoCs
While many CoC requirements stem from contractual obligations rather than explicit regulatory mandates, several frameworks establish conformity documentation requirements that CoCs fulfill.
Frameworks Requiring Conformity Documentation
EU Regulations and Directives: The European Commission requires manufacturers to prepare declarations of conformity for products covered by New Legislative Framework directives. While these are formal declarations rather than supplier CoCs, the underlying conformity documentation often includes supplier certificates.
REACH Substance Restrictions: REACH compliance requires supply chain communication about substances of very high concern (SVHCs). Supplier declarations confirming absence of restricted substances often take CoC form.
RoHS Material Restrictions: RoHS compliance documentation requires technical files demonstrating conformity. Supplier CoCs declaring materials meet RoHS thresholds form part of this documentation. Understanding RoHS exemptions affects CoC content requirements.
Conflict Minerals Disclosure: Conflict minerals compliance under SEC and EU regulations requires supply chain due diligence. Supplier declarations about mineral sourcing often use CoC format. Conflict minerals automation workflows reduce manual CoC processing.
PFAS Restrictions: Emerging PFAS regulations require manufacturers to verify that products do not contain restricted per- and polyfluoroalkyl substances. Supplier CoCs declaring PFAS absence support compliance documentation. PFAS compliance in 2026 will intensify CoC collection requirements.
Proposition 65: California Proposition 65 compliance requires manufacturers to verify products do not contain listed chemicals above safe harbor levels. Supplier CoCs support this verification process.
FDA Requirements: Medical device manufacturers must maintain supplier documentation demonstrating component conformity. While FDA does not mandate specific CoC formats, QSR requirements necessitate documented evidence that suppliers meet specifications.
ISO Standards: Quality management standards like ISO 9001 require organizations to verify that purchased products conform to requirements. Supplier CoCs provide this verification evidence.
Common Certificate of Conformance Template Requirements
Organizations create certificate of conformance templates standardizing information elements and format. Template selection depends on industry, regulatory requirements, and customer specifications.
Standard Template Approaches
Minimal Compliance Templates: Basic templates include only essential elements: supplier/customer identification, product description, specification references, and conformity declaration. These work for low-risk commercial transactions.
Extended Documentation Templates: Comprehensive templates include test result summaries, dimensional verification, visual inspection results, and traceability information. Common in aerospace, medical, and automotive industries.
Regulatory-Specific Templates: Templates designed for specific frameworks like RoHS, REACH, or conflict minerals include framework-specific declarations and attestations. Standardized supplier questionnaire frameworks improve response consistency.
Material-Specific Templates: Templates for specific material categories (metals, plastics, chemicals, electronic components) include relevant specifications and test parameters. Chemical CoCs often include composition data.
Multi-Standard Templates: Combined templates addressing multiple standards simultaneously (e.g., RoHS + REACH + Conflict Minerals) reduce supplier burden and documentation volume.
Template Standardization Challenges
Lack of universal CoC standards creates supplier burden. Large suppliers receive hundreds of different CoC templates from customers, each with unique formats and information requirements. Supplier self-service compliance portals can accept various formats while extracting standardized data.
Industry initiatives like IPC-1752 for materials declaration provide standardization in electronics. However, most industries lack equivalent standards, leaving organizations to create proprietary templates.
Verification and Validation Challenges
Receiving a certificate of conformance does not guarantee actual conformity. Organizations must verify CoC authenticity and validate that documented conformity reflects reality.
Common CoC Verification Challenges
Incomplete Information: CoCs missing essential elements like specification references or product descriptions provide insufficient verification. Incomplete documentation requires follow-up, delaying production.
Generic Declarations: Statements like "meets all applicable standards" or "conforms to customer requirements" lack specificity needed for verification. Effective CoCs cite specific standards with version numbers.
Unsigned or Improperly Authorized: CoCs signed by individuals without proper authority lack legal weight. Supplier qualification should establish authorized signatories.
Outdated Specification References: CoCs referencing obsolete specification versions create ambiguity about conformity. Version control between customers and suppliers prevents this issue.
Contradictory Information: CoCs declaring conformity when accompanying test reports show non-conformances indicate problems with supplier quality systems or documentation integrity.
Lack of Traceability: CoCs that cannot be linked to specific batches, lots, or serial numbers limit their value for root cause analysis when problems arise.
Third-Party Verification Absence: For critical applications, customer verification through inspection or testing provides greater assurance than supplier self-declaration alone.
Validation Best Practices
Organizations should implement supplier risk scoring and ESG ratings to identify high-risk suppliers requiring enhanced verification. Periodic audits of supplier documentation systems validate CoC reliability.
Automated systems can flag incomplete CoCs, identify missing signatures, detect specification mismatches, and track supplier conformance history. AI-native compliance automation reduces manual verification burden.
Risks of Inadequate CoC Documentation
Insufficient certificate of conformance documentation creates operational, legal, and financial risks extending beyond individual transactions.
Primary Risk Categories
Audit Failures: Regulatory audits, customer assessments, and certification body reviews require documented evidence of supplier conformity. Missing or incomplete CoCs result in audit findings, corrective action requirements, and potential certificate suspension.
Product Recalls: When products fail in service, investigators trace failures to source materials. Absent CoCs complicate root cause analysis and may prevent identification of affected product populations. Managing compliance risk proactively includes systematic CoC collection.
Regulatory Non-Compliance: Frameworks requiring conformity documentation impose penalties for inadequate records. Market access depends on demonstrating compliance through documentation.
Liability Exposure: Delivering products that fail to meet specifications exposes organizations to breach of contract claims. CoCs establish that suppliers attested to conformity, shifting liability when materials prove non-conformant.
Supply Chain Disruption: Discovering material non-conformances during production requires emergency material replacement, production stops, and expedited sourcing. Proper CoC verification before material release prevents these disruptions.
Customer Relationship Damage: Delivering products without proper conformity documentation damages customer trust. Lost business from major customers far exceeds the cost of proper CoC management.
Inefficiency and Rework: Chasing missing CoCs after material receipt creates inefficiency. Materials may sit in receiving pending documentation, tying up working capital and delaying production.
How to Manage CoC Documentation at Scale
Organizations receiving hundreds or thousands of CoCs annually cannot rely on manual processes. Systematic approaches to CoC management improve efficiency and reduce compliance risk.
CoC Management Framework Components
1. Standardized Requirements Definition: Establish clear CoC requirements for different material categories, supplier tiers, and risk levels. Document requirements in supplier agreements and purchase order terms.
2. Supplier Communication and Training: Centralized supplier self-service portals provide suppliers with templates, instructions, and submission interfaces. Training reduces incomplete submissions.
3. Automated Receipt and Processing: Electronic submission systems automatically route CoCs to appropriate reviewers, extract key data elements, and flag incomplete submissions. Integration with ERP systems links CoCs to purchase orders and material receipts.
4. Verification Workflow Management: Automated workflows ensure appropriate personnel review CoCs before material release. Verification steps include completeness checks, specification matching, and signature validation.
5. Digital Archive and Retrieval: Searchable digital repositories enable rapid CoC retrieval during audits, investigations, or customer requests. Continuous audit-ready documentation requires organized archiving.
6. Exception Management: Track CoCs with deviations, non-conformances, or missing information. Escalation procedures ensure timely resolution before materials enter production.
7. Supplier Performance Tracking: Monitor supplier CoC submission timeliness, completeness, and accuracy. Use performance metrics in supplier evaluations and sourcing decisions.
8. Integration with Quality Systems: Link CoCs to material inspection records, non-conformance reports, and corrective action systems. This integration supports root cause analysis when quality issues arise.
Scaling Challenges
Manual CoC management fails as organizations grow supplier bases, expand product portfolios, or increase production volumes. Replacing spreadsheets with scalable systems becomes necessary as transaction volumes exceed manual processing capacity.
Multi-plant organizations require standardized approaches across facilities. Standardizing compliance across plants and regions prevents inconsistent CoC requirements and documentation practices.
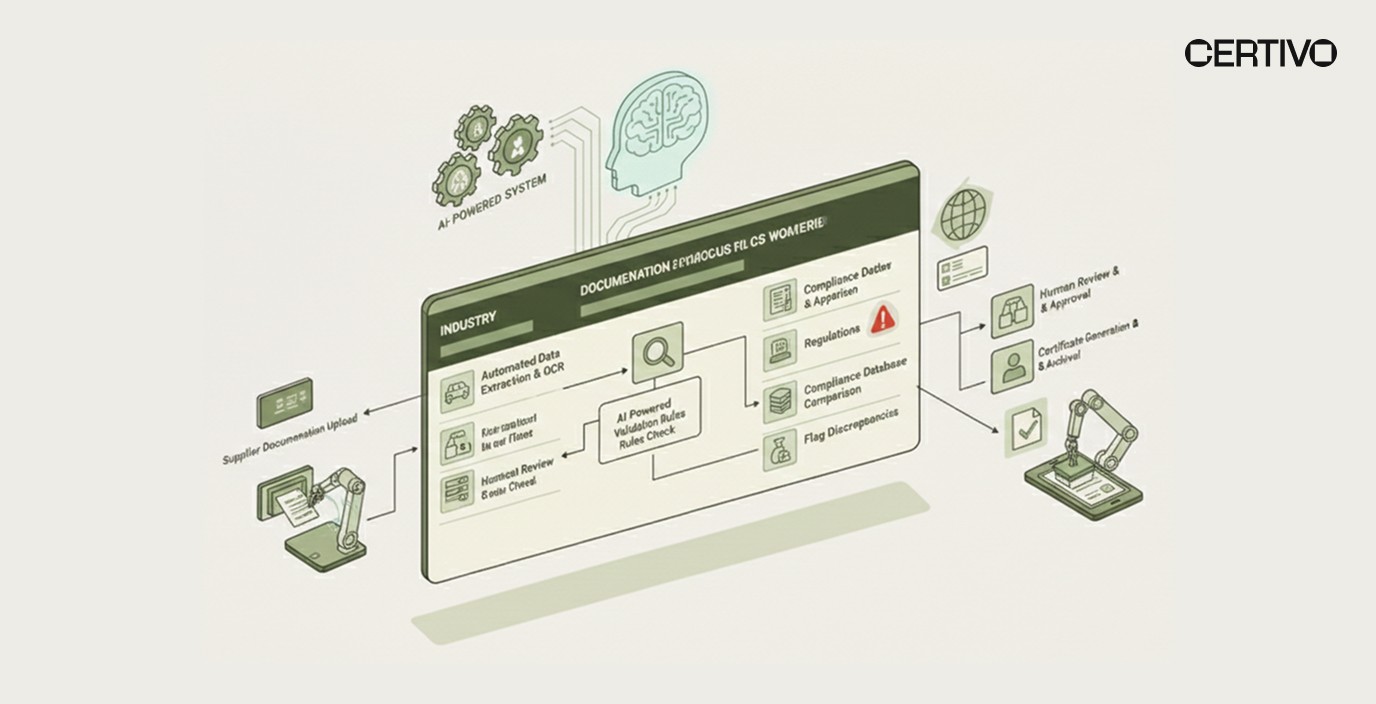
AI-Native Automation for Certificate Management
AI-native compliance automation transforms certificate of conformance management from administrative burden to strategic capability. Machine learning, natural language processing, and intelligent automation reduce manual effort while improving accuracy.
AI Capabilities in CoC Management
Automated Data Extraction: AI systems extract key information from CoC documents regardless of format. Optical character recognition (OCR) processes scanned documents. Natural language processing identifies product descriptions, specifications, and declarations even in non-standardized formats.
Intelligent Completeness Verification: CORA-powered regulatory intelligence validates that CoCs contain all required elements based on material category, regulatory requirements, and contractual obligations. Missing information triggers automated supplier follow-up.
Specification Matching and Validation: AI compares CoC content against purchase order specifications, technical drawings, and material standards. Automated matching identifies discrepancies before material release.
Multi-Framework Cross-Referencing: When CoCs support multiple regulatory frameworks (REACH, RoHS, PFAS), AI systems verify that declarations address all applicable requirements. CORA intelligence identifies gaps in regulatory coverage.
Traceability Linking: AI automatically links CoCs to purchase orders, material receipts, inspection records, and downstream products. This enables rapid impact analysis when supplier issues arise.
Anomaly Detection: Machine learning identifies unusual patterns suggesting CoC problems: signatures that don't match previous submissions, test results inconsistent with material types, or specification references that don't exist.
Automated Customer Response: When customers request conformity documentation, AI retrieves relevant CoCs, compiles compliance summaries, and generates response packages. Customer trust centers and self-service reporting enable customer self-service access.
Predictive Supplier Risk Scoring: CORA-driven compliance intelligence analyzes supplier CoC history to identify reliability trends. Suppliers with frequent incomplete submissions or conformity issues receive higher risk scores, triggering enhanced verification.
Regulatory Change Impact Analysis: When regulations change, AI identifies which suppliers and materials require updated CoC language. Automated notifications inform suppliers of new requirements.
From Administrative Burden to Strategic Asset
AI automation transforms CoC management from cost center to strategic capability. Organizations gain:
Faster material release through automated verification
Reduced compliance risk via comprehensive validation
Lower operational cost by eliminating manual processing
Better supplier performance through automated tracking
Audit readiness via organized digital archives
Customer satisfaction through rapid documentation response
AI in supply chain compliance management represents fundamental operational transformation rather than incremental improvement.
Frequently Asked Questions
What is the difference between a certificate of conformance and a certificate of analysis?
A certificate of conformance (CoC) is a supplier's declaration that products meet specified requirements, while a certificate of analysis (CoA) provides detailed test results and analytical data. CoCs attest to conformity; CoAs document measurements and test outcomes. Many transactions require both documents—the CoC provides the conformity declaration while the CoA supplies supporting test evidence.
Is a certificate of conformance legally required?
Legal requirements for CoCs vary by industry, product category, and jurisdiction. While many CoC requirements stem from contractual obligations rather than explicit regulations, several frameworks establish conformity documentation requirements. Medical device regulations, aerospace standards, and construction codes often mandate documented conformity evidence. Organizations should identify applicable requirements based on their specific products and markets.
How long should certificates of conformance be retained?
Retention requirements depend on regulatory frameworks, customer contracts, and product liability considerations. Medical device manufacturers typically retain CoCs for the product lifetime plus 7-10 years. Aerospace and defense industries maintain permanent records. Construction materials often require retention matching structure lifetime. Organizations should establish retention policies addressing regulatory minimums, contractual obligations, and liability exposure. Continuous audit-ready documentation requires organized long-term archiving.
Can a certificate of conformance be generated electronically?
Electronic CoCs are acceptable in most applications if they include valid electronic signatures meeting applicable standards. Digital signatures provide authentication, non-repudiation, and integrity verification. Electronic CoC systems should include access controls, audit trails, and backup procedures. Some customer-specific requirements or regulatory frameworks may specify paper documentation, so organizations should verify acceptance before implementing fully electronic systems.
How can manufacturers automate certificate of conformance collection and verification?
Automation begins with supplier self-service portals providing standardized submission interfaces. AI-powered systems extract data from submitted CoCs regardless of format. CORA-driven compliance intelligence validates completeness, matches specifications, and flags anomalies. Integration with ERP and PLM systems links CoCs to purchase orders and material receipts. Automated workflows route CoCs for review and archive approved documents in searchable repositories. Certivo's AI-native platform provides end-to-end CoC management capabilities.
What should be included in a certificate of conformance template?
Essential CoC template elements include: supplier and customer identification, purchase order reference, product description with part numbers, specification or standard references, conformity declaration statement, date, and authorized signature. Additional valuable elements include test report references, batch/lot numbers, manufacturing date, inspection results, and deviation notes. Template content should match industry requirements, regulatory frameworks, and customer specifications. Standardized supplier questionnaire frameworks reduce supplier burden while ensuring complete documentation.
Conclusion
Certificate of conformance documentation has evolved from administrative formality to strategic compliance requirement. As regulatory frameworks expand substance restrictions, customers demand verified conformity, and supply chains span multiple tiers, systematic CoC management determines operational efficiency and compliance risk.
Organizations relying on manual CoC collection, spreadsheet tracking, and paper archives face scalability limitations as transaction volumes grow. The shift to AI-native compliance automation enables extraction of key information from any format, intelligent verification against specifications and regulations, automated supplier follow-up for incomplete submissions, and rapid retrieval during audits or customer requests.
Understanding CoC requirements, distinctions from other certificates, verification best practices, and automation strategies positions organizations for efficient compliance management across expanding regulatory landscapes. Explore how Certivo's AI-powered platform automates certificate of conformance management while maintaining continuous audit-ready documentation.
Kunal Chopra
Kunal Chopra is the CEO of Certivo, an AI-driven compliance management platform revolutionizing how manufacturers navigate regulatory challenges. With a career spanning over two decades, Kunal is a seasoned technology leader, 3x tech CEO, product innovator, and board member with a passion for driving transformative growth and innovation.
Before leading Certivo, Kunal spearheaded successful transformations at renowned companies like Beckett Collectibles, Kaspien, Amazon, and Microsoft. His strategic vision and operational excellence have led to achievements such as a 25x EBITDA valuation increase at Beckett Collectibles and a 450% shareholder return at Kaspien. He has a track record of turning challenges into opportunities, delivering operational efficiencies, and driving market expansions.
Kunal’s deep expertise lies in blending technology and business strategy to create scalable solutions. At Certivo, he applies this expertise to empower manufacturers, using AI to turn product compliance from an operational burden into a strategic advantage.
Kunal holds an MBA from The University of Chicago Booth School of Business, an MS in Computer Science from Clemson University, and a BE in Computer Engineering from The University of Mumbai. When he’s not transforming businesses, Kunal is an advocate for innovation, growth, and building cultures that inspire excellence.
Stay tuned for insights from Kunal on how technology can redefine compliance, drive efficiency, and create opportunities for growth in the manufacturing sector.